
SOLVED Which of the following, if any, contain nonpolar covalent bonds
In this article, we will discuss NF3 lewis dot structure, molecular geometry or VSEPR shape, bond angle, hybridization, etc. We will also discuss is NF3 polar or non-polar? NF3 is used to make chemicals or also used as a component of rocket fuels. It is a strong greenhouse gas and it has the potential to reduce global warming greater than co2.

BF3 and NF3 both are covalent compounds but NF3 is polar whereas BF3 is
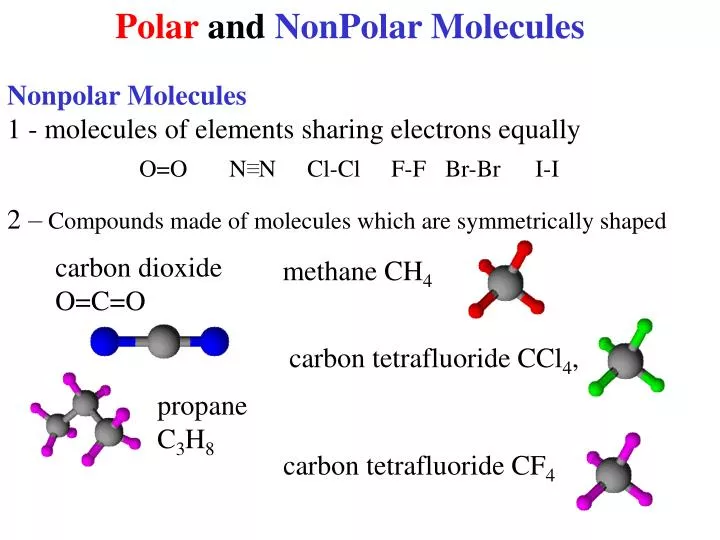
In a nonpolar covalent bond, the distribution of electrical charge is balanced between the two atoms. Figure 4.3.2 4.3. 2: A nonpolar covalent bond is one in which the distribution of electron density between the two atoms is equal. The two chlorine atoms share the pair of electrons in the single covalent bond equally, and the electron density.

Answered 5. How many of the following contain at… bartleby
In this blog post, we will explore the Lewis structure for NF3, its bond angle, and whether it is polar or nonpolar. We will also answer questions like how many bonds NF3 has and dive into its molecular geometry. So, let's dive in and unravel the mysteries behind NF3! Lewis Structure for NF3: A Molecular Dance of Nitrogen and Fluorine

Is NF3 Polar or Nonpolar (Nitrogen Trifluoride) YouTube
In a polar covalent bond, sometimes simply called a polar bond, the distribution of shared electrons within the molecule is no longer symmetrical (see figure below). Figure 5.3.4 5.3. 4: In the polar covalent bond of HF HF, the electron density is unevenly distributed. There is a higher density (red) near the fluorine atom, and a lower density.

Nitơ trifluoride NF3 khí đặc biệt cho Plasma
The nitrogen trifluoride (NF3) is a polar covalent bond because the electrons are shared between fluorine and nitrogen. Let suppose if the two fluorine atoms come together, they will make a nonpolar covalent bond. Also, if the two nitrogen atoms share the electrons equally, it will make nonpolar covalent bonds.
Ch4 Polar Or Nonpolar / What Is A Nonpolar Covalent Bond? Science
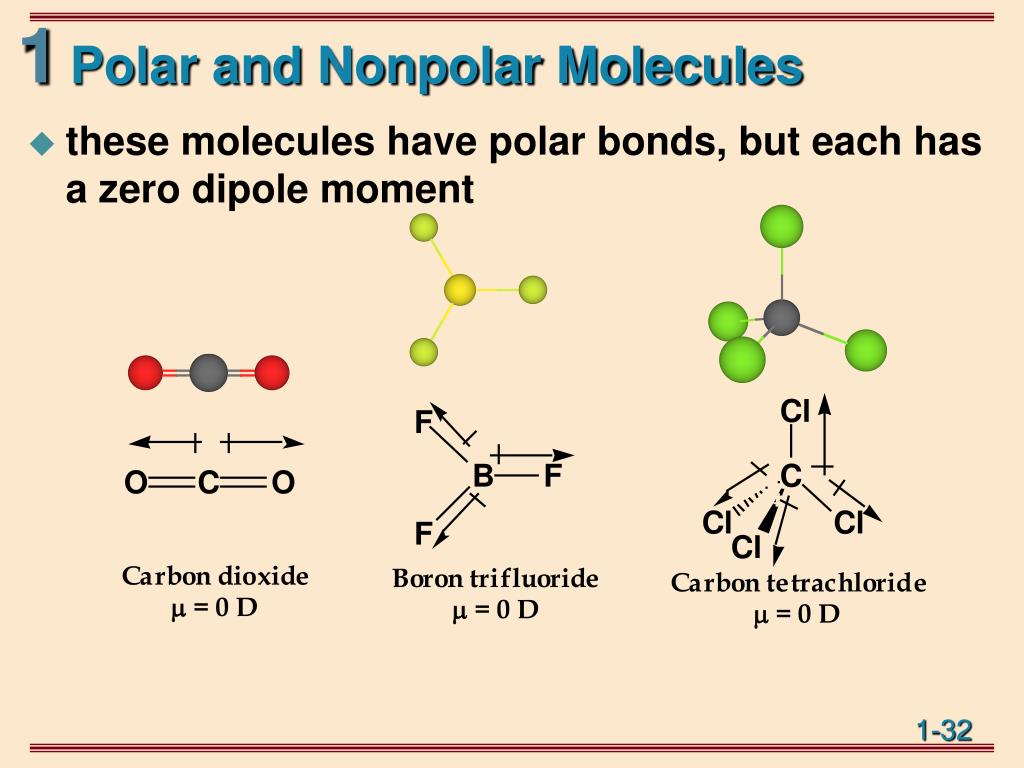
BF3 (Boron Trifluoride): This molecule is nonpolar. Boron and Fluorine have different electronegativities, but the shape of BF3 is trigonal planar, which allows for the dipoles to cancel out. Answer. 5. BrCl3 (Bromine Trichloride): This molecule is polar. Bromine and Chlorine have different electronegativities, creating a dipole moment.

BF3 and nf3 both molecules are covalent BF3 is nonpolar and nf3 is
NF3 is a chemical formula for Nitrogen Trifluoride. It is used in manufacturing semiconductors. Here in this video, we share a detailed step-by-step method t.

Bf3 Polar or Nonpolar JamisonecWells
NF3 is a covalent (polar covalent) compound because when one nonmetal combines with another nonmetal, it usually forms a covalent compound. Here, N is a nonmetal and F is also a nonmetal. So when they combine, it forms a covalent compound. Well, now you have got to know that NF3 is a covalent compound, but let me explain the in-depth reason why.
Ppt Polar Bonds And Molecules Powerpoint Presentation, Free Download 587
Now in the next step we have to check whether these N-F bonds are polar or nonpolar. And we also have to check the molecular geometry of NF3. Step #2: Check whether individual bonds are polar or nonpolar. The chemical bonds can be either nonpolar, polar or ionic depending on the difference of the electronegativity values (ΔEN) between the two.

Is NF3 Polar or Nonpolar? Techiescientist
The molecules is perfectly symmetrical, so every electron pair on each fluorine cancels out the electron pairs of every other fluorine. For this reason, this molecule is non-polar. In CHFX3 C H F X 3, however, the hydrogen does not have 3 other electron clouds around it like the fluorine do.

Bf3 and nf3 both molecules are covalent but bf3 is non polar and nf3 is
When you place a molecule with an electric dipole in an electric field, a force acts to turn the molecule so that the positive and negative ends line up with the field. The magnitude of the turning force is given by the formula. µ = q × d. where q is the amount of charge and d is the distance between the two charges. µ is the turning moment.

Is NF3 (Nitrogen trifluoride) Polar or NonPolar? YouTube
Molecular Polarity. To determine if a molecule is polar or nonpolar, it is frequently useful to look at Lewis structures. Nonpolar compounds will be symmetric, meaning all of the sides around the central atom are identical - bonded to the same element with no unshared pairs of electrons.Polar molecules are asymmetric, either containing lone pairs of electrons on a central atom or having atoms.

Is CH3F Polar or Nonpolar? Techiescientist
DO NOT FORGET TO SUBSCRIBE!LinkedIn: https://www.linkedin.com/in/kevan-j-e.Snapchat: https://www.snapchat.com/add/kravonoInstagram: https://www.instagram.c.

Lectura Enlaces covalentes Biología I Association LEA
The molecule is symmetric. The two oxygen atoms pull on the electrons by exactly the same amount. Propane is nonpolar, because it is symmetric, with H atoms bonded to every side around the central atoms and no unshared pairs of electrons. Exercise 4.12. 1. Label each of the following as polar or nonpolar.
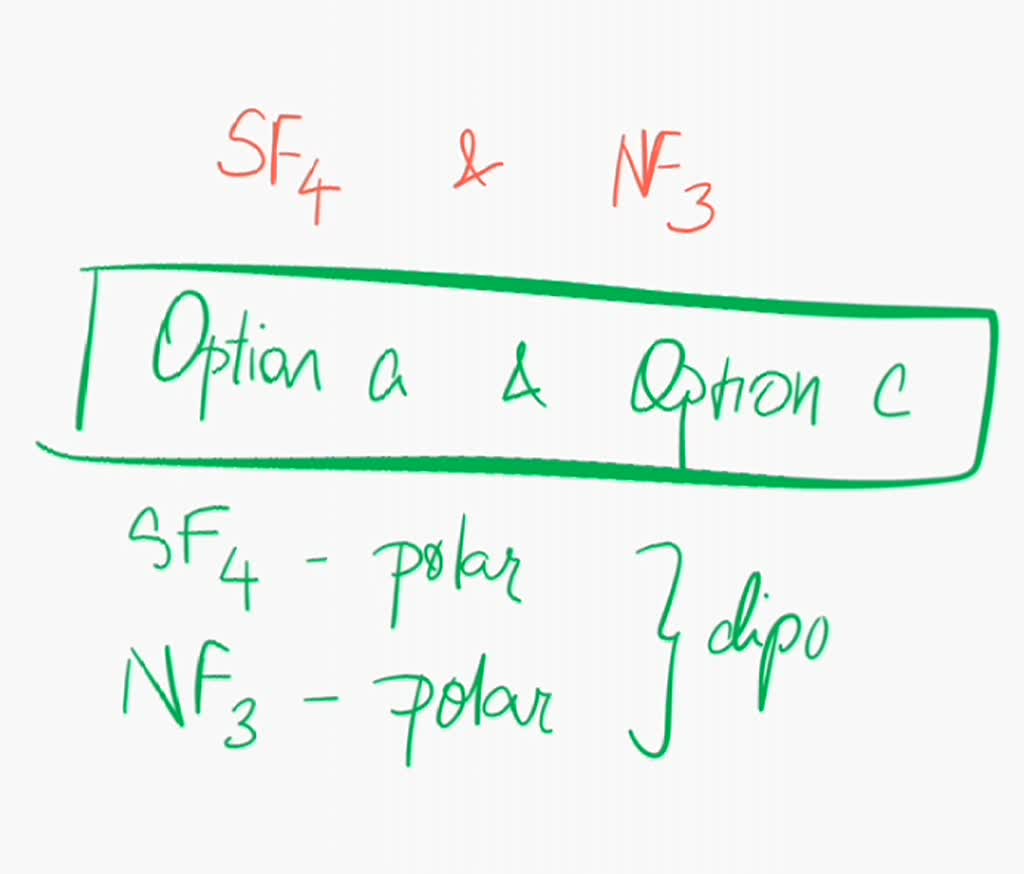
SOLVED Are sulfur tetrafluoride (SF4) and nitrogen trifluoride (NF3
You'll get a detailed solution from a subject matter expert that helps you learn core concepts. Question: Compare and contrast NH3 and NF3. Are they polar or nonpolar compounds? What type of intermolecular force is present for each compound? Which compound has the higher boiling point?

BF3 and nf3 both molecules are covalent BF3 is nonpolar and nf3 is
High reactivity: Nf3 is highly reactive and can easily react with various substances, including water, metals, and organic compounds. Non-polarity: Nf3 is a non-polar molecule due to the symmetry in the arrangement of its atoms. This characteristic affects its behavior in different chemical reactions.